Protein Conformation & Kinetic Analysis by HDX MS
Background & Overview
Hydrogen-deuterium exchange mass spectrometry(HDX MS) is very useful in the study of many aspects of proteins, especially the analysis of protein conformation and dynamics. These results have important guiding significance for a deep understanding of the physiological activities and applications of proteins. BOC Sciences uses nano-scale color fillers, which can greatly shorten the time required for liquid phase analysis without losing the chromatographic separation effect, and has an ultra-high resolution. In addition, we also adopted a high-resolution mass spectrometry system, which not only ensures the reliability of the experimental data, but also is more direct, fast and routine.
Analysis Process
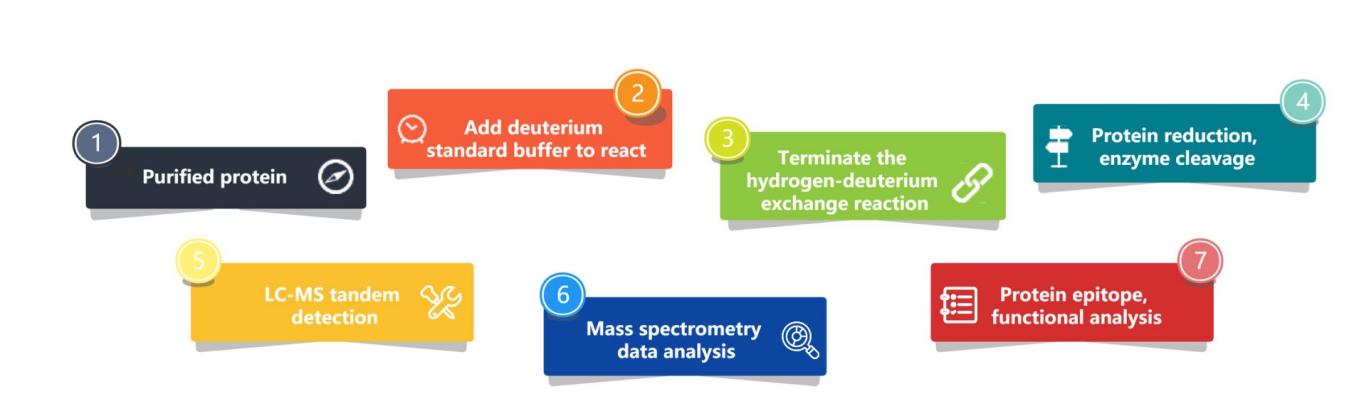
Our analysis principle is that by placing the protein in a deuterated solution, the hydrogen atoms on the surface of the protein are exchanged with the deuterium atoms in the solvent, and the exchanged protein is digested to produce polypeptide fragments. Finally, mass spectrometry was used to identify the mass of the peptides (peptides located on the surface of the protein are more likely to undergo hydrogen tritium atom exchange and increase in mass than those located in the interior of the protein, thereby inferring the protein epitope conformation).
Our Services
Protein folding research is one of the core research fields of molecular biology today, with important theoretical and practical significance. Its main content is to study the law, stability and relationship between the specific three-dimensional spatial structure of protein and its biological activity. BOC Sciences is able to reveal which parts are folded and when by monitoring changes in deuteration during folding, as well as what factors may influence folding and various folding states. In addition to the study of the folding and unfolding of individual proteins, our technology supports understanding how other proteins participate in folding or maintain a folded state.
Understanding the structure of a protein is an important factor in understanding its function. Our hydrogen-deuterium exchange mass spectrometry played an important role in this work. Our research objects cover two categories of virus-related proteins and virus-unrelated proteins. Not only that, but we were able to combine hydrogen-deuterium exchange mass spectrometry with structural analysis from cryo-EM, modeling, and other tools to provide more information. Our services include but are not limited to, the analysis of multidomain proteins, quaternary interactions, and protein evolution relationships.
The activity and function of proteins are often regulated by binding to other molecules; therefore, understanding these events can provide important insights into protein function and disease-related states. We use hydrogen-deuterium exchange mass spectrometry to study protein interactions including antigen/antibody, protein/macromolecule, and protein/small molecule.
Technical Advantages

Sample Request
- Protein-ligand and protein samples
- Sample concentration >1ug/ul
- Sample size >10ug
- NOTE: Samples are ideally free of detergents.
If you are interested in our services, please contact us immediately, then fill in the complete inquiry form, and we will reply to you as soon as possible.
Reference
- Pirrone G F, et al. Applications of hydrogen/deuterium exchange MS from 2012 to 2014. Analytical chemistry. 2015, 87(1): 99-118.





