Deuteration of Silanes Based on DCL™
How Much Do You Know About Deuterated Silane?
Isotopically labeled compounds are now more and more favored by scientists, because their application scope gradually penetrates into various fields, such as life science, food and drug, medicine, agriculture, environment, geology and so on. In the field of chemistry, isotope-labeled compounds can be used as mechanical probes and as internal standards for mass spectrometry research; in the field of life sciences, isotope-labeled compounds play a key role in applications such as metabolomics, proteomics, and clinical research; In the medical field, isotopically labeled compounds are critical to the drug development process and can be used as diagnostic tools in medical research.
Deuterium labeling of compounds is an important practice common to the pharmaceutical industry. In addition to common C-H bonds, Si-H bonds can also be used for deuterium substitution. Deuterated silanes refer to compounds obtained by replacing hydrogen in silanes with deuterium isotopes. They can not only be used as important probes for the study of organosilicon chemical mechanisms, but also as general reagents for deuterium labeling. Both SiEt3D, SiMe2PhD and SiPh2D2 are common deuterated silanes.
Typically, deuterated silanes are prepared by reduction of halosilanes with LiAlD4 or NaBD4 or by noble metal transition metal-catalyzed isotopic exchange of Si-H/D bonds between silanes and D2. However, the use of expensive metal deuteride reagents and the equipment-demanding D2 undermine their widespread use in synthetic chemistry. Therefore, the development of mild, sustainable, and cost-effective strategies for deuteration of silanes is an important research direction.
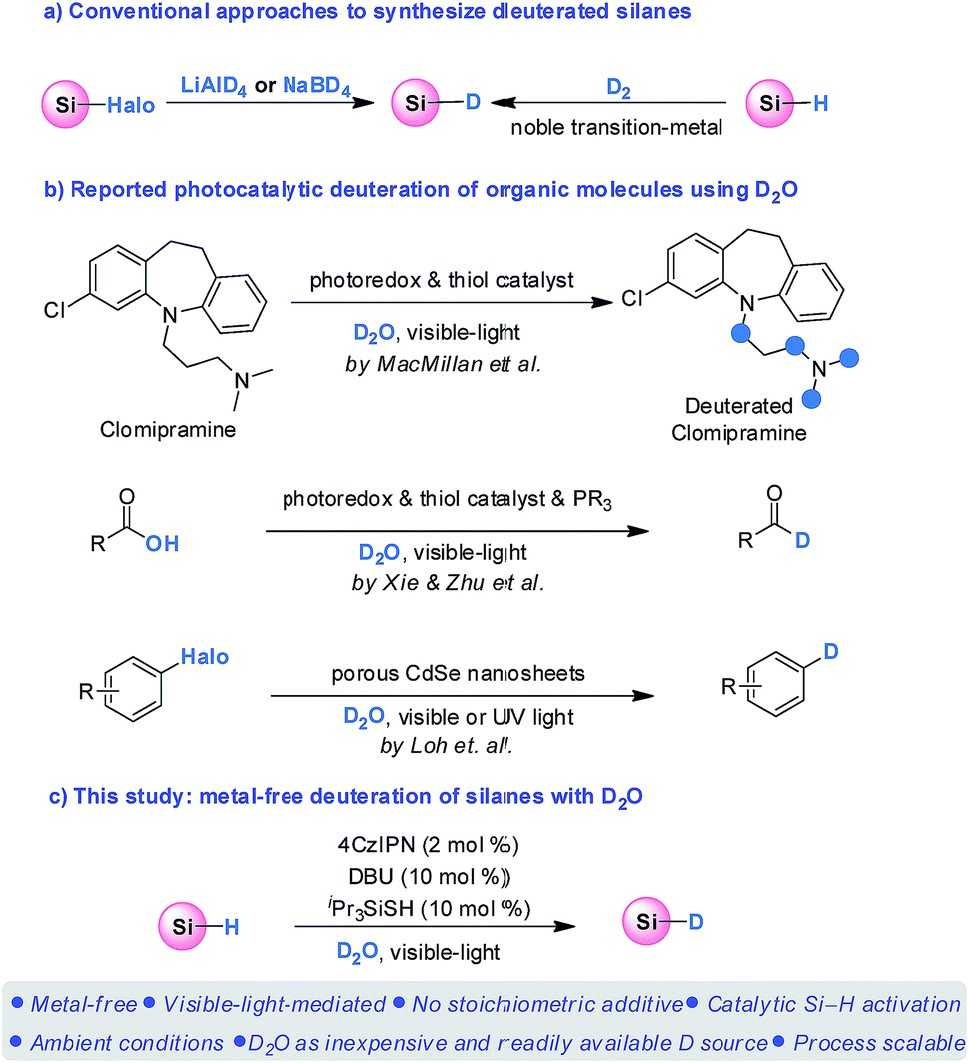 Fig.1 Silane deuteration and photocatalytic deuteration using D2O.[1]
Fig.1 Silane deuteration and photocatalytic deuteration using D2O.[1]
The Role of Deuterated Silanes
To discuss the role of deuterated silanes, start with hydridosilanes. Hydrosilane is a kind of environmentally friendly reducing agent, which is widely used in chemical synthesis and can be used to reduce C-X (X=C, N, O) multiple bonds. In addition, hydrosilanes can be used to catalyze the reduction of carbon-halogen bonds, including non-reactive C-F bonds. Deuterated silanes can also undergo hydrosilylation reactions, while allowing further introduction of more deuterium labels. In conclusion, deuterated silanes show great potential to be used as isotope labeling reagents.
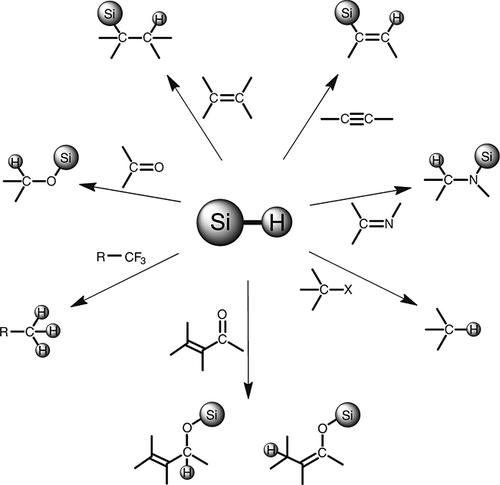 Fig.2 Some synthetic applications of hydrosilanes.[2]
Fig.2 Some synthetic applications of hydrosilanes.[2]
DCL™ in Deuteration of Silanes
Our DCL™ technology platform provides an efficient catalytic procedure for the synthesis of deuterated silanes that is simple to operate, mild reaction conditions, green solvents, and inexpensive. Our method is applicable to a wide variety of silanes, resulting in excellent D-incorporated products. Not only that, our platform enables the hydrosilylation of multiple bonds such as C-O and C-N as well as a variety of ketones and aldehydes, incorporating D into a variety of organic molecules. In conclusion, our platform is expected to facilitate the widespread use of these important reagents, deuterated silanes, in both academic and industrial settings.
Our DCL™ technology platform is designed to help customers and partners complete the selective deuteration of various compound structures, always escorting your research and development projects.
References
- Rong Zhou, et al. Visible-light-mediated deuteration of silanes with deuterium oxide. Chemical Science. 2019.
- Jesús Campos, et al. Large-scale preparation and labelling reactions of deuterated silanes. Journal of Labelled Compounds & Radiopharmaceuticals. 2012, 55(1):29-38.
Customer Intellectual Property Protection
BOC Sciences has always regarded intellectual property as the most valuable asset of the company and its customers. We have signed non-disclosure agreements with customers and employees before the project starts, and provide synthetic route design and synthesis services in strict accordance with the terms of the non-disclosure agreement, striving to provide customers with target compounds in the shortest time possible.

 Fig.1 Silane deuteration and photocatalytic deuteration using D2O.[1]
Fig.1 Silane deuteration and photocatalytic deuteration using D2O.[1] Fig.2 Some synthetic applications of hydrosilanes.[2]
Fig.2 Some synthetic applications of hydrosilanes.[2]

