Deuteration of Arenes Based on DCL™
Background & Overview
In recent years, the incorporation of hydrogen atom isotopes into organic molecules has received considerable attention, and as more and more deuterated molecules are marketed as new drugs, there has been an increased interest in the development of convenient and robust deuterated compounds. Typically, deuterium isotope labeling can be used to understand the metabolism of active drugs and identify specific metabolites. They are also well suited for determining kinetic isotope effects (KIEs) in fundamental mechanistic studies. Not only that, but deuterium-labeled compounds are also a major source of internal standards for the preparation of liquid chromatography-mass spectrometry (LC-MS) analysis.
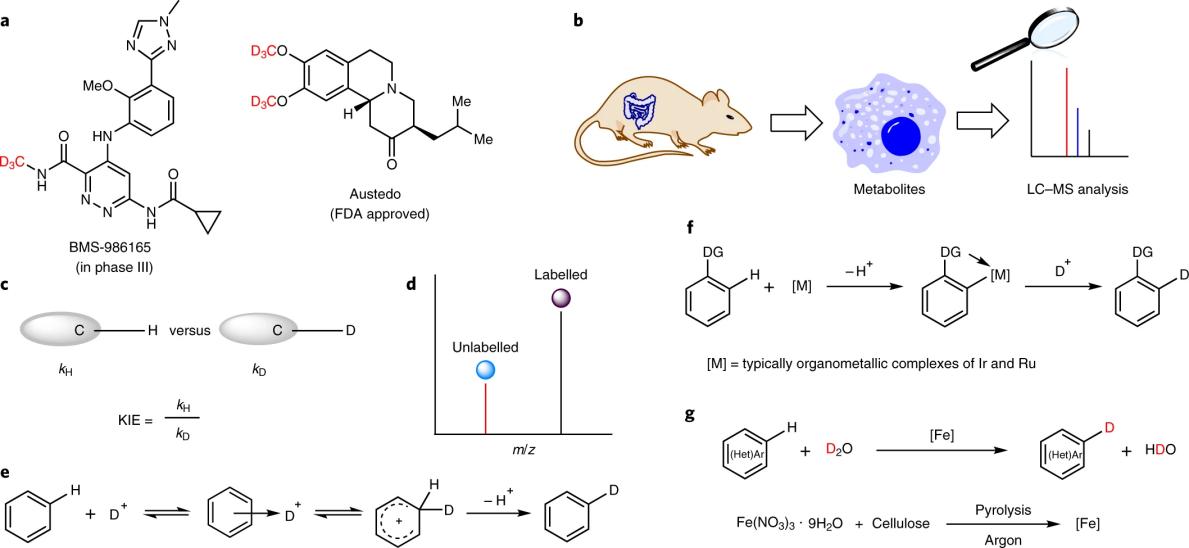 Fig.1 Applications of deuteration and methods for H/D exchange.[2]
Fig.1 Applications of deuteration and methods for H/D exchange.[2]
Aromatic hydrocarbons are found in bioactive molecules and related compounds such as pharmaceuticals, natural products, agrochemicals, etc. Incorporation of deuterium into complex aromatic scaffolds may enhance their chemical properties, half-life, and reduce their possible metabolic degradation. We therefore constructed a method for deuteration of aromatic hydrocarbons that allows efficient incorporation of deuterium isotopes into bioactive molecules after synthesis.
Existing Strategy for the Synthesis of Deuterated Arenes
The acid-mediated hydrogen-deuterium exchange reaction is one of the oldest known methods for labeling aromatic hydrocarbons. However, they only allow the selective incorporation of deuterium for simple substrates according to the electrophilic aromatic substitution mechanism. Furthermore, the necessity of using high temperature and concentrated strong acid for this method leads to poor functional group tolerance and greater safety risk, especially on large scale synthesis.
Based on advances in homogeneous metal-catalyzed C–H activation, a variety of organometallic complexes have been developed for catalytic H/D exchange reactions of arenes, as well as aliphatic amines at the α or β position. For example, homogeneous iridium-based Crabtree and Kerr catalysts have been used for C(sp2)-H hydrogen isotope exchange reactions using D2 gas. In addition to the identified organometallic complexes, photocatalysts and heterogeneous materials have also been investigated in labeling reactions.
Unfortunately, in all of these cases, the selectivity and tolerance of readily reducible functional groups and halogens are challenging. Furthermore, most deuterated heterogeneous catalysts rely on expensive precious metals, which hinders their use in agrochemical, pharmaceutical or food industries.
DCL™ in Deuteration of Arenes
For deuteration of aromatic hydrocarbons, our DCL™ technology platform explores a broad substrate range, enabling excellent chemical and regioselective deuteration under relatively mild conditions, enabling broader functional group tolerance.
Our capabilities are shown below:
Notably, our platform can further complete the late deuteration of arenes in some representative drugs and natural products. Specifically include melatonin, N-acetyl serotonin, nucleoside analog kinetin, inosine, pentoxifylline, alkaloids, tyrosol, resveratrol, thymol, arbutin, dropromazine, DL- Aminoglutamine, carvacrol and estradiol, etc.
References
- Farizyan M, et al. Palladium-Catalyzed Nondirected Late-Stage C–H Deuteration of Arenes. Journal of the American Chemical Society. 2021, 143(40): 16370-16376.
- Li W, et al. Scalable and selective deuteration of (hetero) arenes. Nature chemistry. 2022, 14(3): 334-341.
Customer Intellectual Property Protection
BOC Sciences has always regarded intellectual property as the most valuable asset of the company and its customers. We have signed non-disclosure agreements with customers and employees before the project starts, and provide synthetic route design and synthesis services in strict accordance with the terms of the non-disclosure agreement, striving to provide customers with target compounds in the shortest time possible.

 Fig.1 Applications of deuteration and methods for H/D exchange.[2]
Fig.1 Applications of deuteration and methods for H/D exchange.[2]


