Deuteration of Nucleosides and (oligo)nucleotides Based on DCL™
Background & Overview

Nucleosides and (oligo)nucleotides, as important components of living organisms, are widely present in various organisms. They are involved in many biological processes, such as the transmission of genetic information, energy processes or cellular communication. Therefore, a large number of nucleoside and (oligo)nucleotide derivatives have been found to be effective drugs in the treatment of major public health problems such as HIV, asthma or cancer. Not only that, but deuterated nucleotides are widely used in biochemistry and molecular biology to suppress unnecessary proton resonances in NMR structural studies, as well as in atom transfer experiments for DNA damaging agents.
Recently, modified nucleic acids are widely used as suitable tools in molecular biology, biotechnology and medicine, for example, as an experimental tool to inhibit gene expression for the development of high-potential therapeutic drugs. Due to the structural complexity of nucleoside and (oligo)nucleotide derivatives, selective chemical transformation of these molecules is an important challenge. Among them, the development of efficient and selective hydrogen isotope exchange reactions for their late deuterium and tritium labeling is crucial.
In fact, tritiated analogs of drug candidates are important tools for studying drug absorption, distribution, metabolism, and excretion in vivo. Deuterated molecules are also widely used in various life science fields, such as metabolomics and proteomics. Therefore, based on our advanced DCL™ technology platform, we can help you to complete the synthesis challenges of deuterated nucleosides and (oligo)nucleotides.
Existing Strategy for the Synthesis of Nucleosides & (oligo)nucleotides
Due to the complexity and relative instability of nucleosides and (oligo)nucleotides, conventional hydrogen isotope labeling is mainly achieved by multi-step synthesis, starting from precursors such as halogen, ketone or aldehyde derivatives, and then catalyzed using deuterium gas Dehalogenation or reduction with labelling reagents such as NaBD4 or LiAlT4. Acid-base exchange or Pd/C-catalyzed H/D exchange methods in deuterated water based on purine derivatives have also been reported. However, due to the high risk factor of tritiated water, such tritium labeling methods are not particularly suitable. Later, a broad-spectrum method for deuterium labeling of purine substructures using ruthenium nanoparticles (RuNp) as catalysts, and deuterium and tritium gases as isotope sources, respectively, appeared, as shown in the following figure:
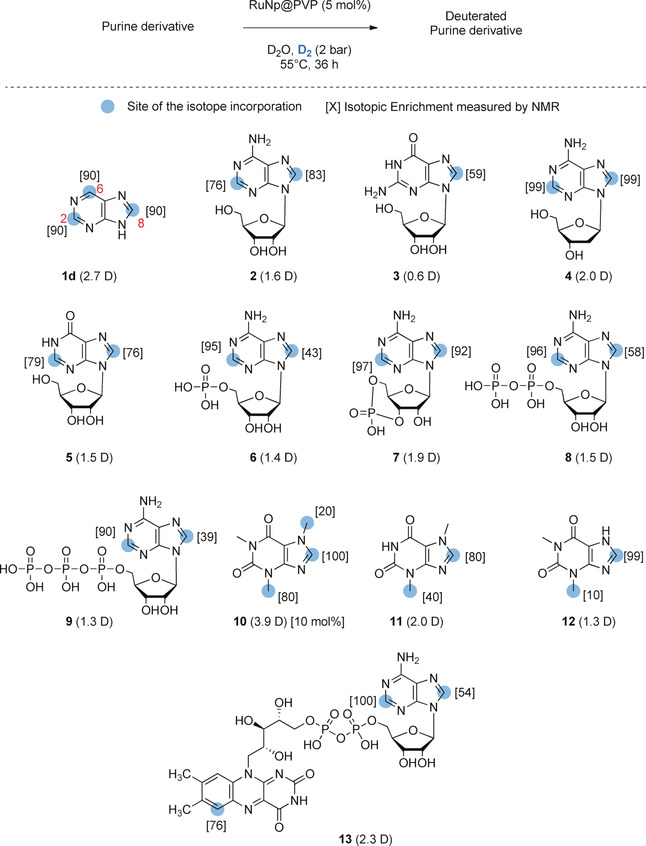 Fig.1 RuNp-catalyzed deuterium labelling of representative purine derivatives.[1]
Fig.1 RuNp-catalyzed deuterium labelling of representative purine derivatives.[1]
DCL™ in Undirected Deuteration Nucleosides & (oligo)nucleotides
Our DCL™ technology platform is based on a novel approach that allows for the regioselective incorporation of hydrogen isotopes into complex biologically relevant substrates, including drugs and biomolecules, under mild reaction conditions. In addition, our technology enables the synthesis of deuterated oligonucleotide internal standards for quantitative LC-MS analysis. Our substrate range includes purine and pyrimidine based natural substrates such as nucleosides, nucleotides and analogs.
Platform Advantage
- Efficient and convenient general method
- Mild reaction conditions
- Broad substrate range
- Broad solvent tolerance
Reference
- Palazzolo A, et al. Efficient access to deuterated and tritiated nucleobase pharmaceuticals and oligonucleotides using hydrogen-isotope exchange. Angewandte Chemie. 2019, 131(15): 4945-4949.
Customer Intellectual Property Protection
BOC Sciences has always regarded intellectual property as the most valuable asset of the company and its customers. We have signed non-disclosure agreements with customers and employees before the project starts, and provide synthetic route design and synthesis services in strict accordance with the terms of the non-disclosure agreement, striving to provide customers with target compounds in the shortest time possible.


 Fig.1 RuNp-catalyzed deuterium labelling of representative purine derivatives.[1]
Fig.1 RuNp-catalyzed deuterium labelling of representative purine derivatives.[1]

