Deuteration of Carbohydrates Based on DCL™
Background & Overview
Deuterium labeling (replacing one or more hydrogens in a molecule with deuterium) of organic compounds is gaining popularity in drug design after the discovery of several D-labeled drugs with better pharmacokinetic properties. Deuterium is a stable, safe, and readily available hydrogen isotope with nearly identical chemical reactivity and a low natural abundance of<0.02% in water. Essential for both reaction pathway studies and analytical methods.
Carbohydrates are the most abundant biomolecules and play an integral role in a wide range of biological processes, including cell-cell recognition, protein folding, inflammation and infection. Therefore, the possibility of selectively functionalizing carbohydrates to enhance the physiological properties of the parent molecule is very attractive. We have developed deuterated technology for various types of carbohydrates to meet a wide range of customer needs.
Application of Deuterated Peptides
Deuterated carbohydrates play important roles in biosynthesis, the study of metabolic pathways, the simplification of complex NMR spectra, and the study of glycolipid dynamics in cell membranes.
Biosynthesis & metabolic pathway research
Understanding and probing small molecule uptake in cells is challenging and requires spatially large chemical labels or radioisotopes. Deuterated carbohydrate probes can serve as "label-free" substrates to assess the uptake of these carbohydrates by the non-pathogenic model organism Mycobacterium smegmatis, by nuclear reaction assays. This is of particular importance for the development of new therapeutic and diagnostic approaches and for exploring carbohydrate uptake in a range of biotechnological and medically related fields.
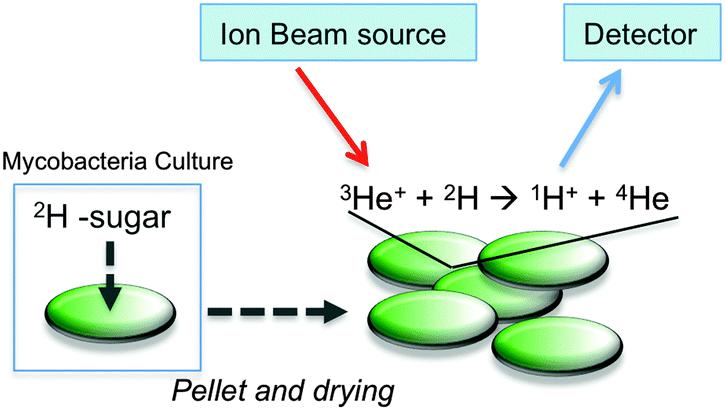 Fig.1 Schematic illustration for assessing 2H-carbohydrate uptake by M. smegmatis and ion beam analysis.[2]
Fig.1 Schematic illustration for assessing 2H-carbohydrate uptake by M. smegmatis and ion beam analysis.[2]
As another example, deuterated bacterial cellulose can be used as a model substrate to understand the role of cellulose structure in enzymatic hydrolysis or other complex transformations.
Simplification of complex NMR spectra
NMR spectroscopic analysis of carbohydrates is often affected by severe overlap of resonance signals, especially in 1H NMR spectra. Selective deuterium labeling is a method to simplify 1H NMR spectroscopy. The spectral simplification resulting from selective deuteration provides additional information for conformational analysis, which is particularly useful for investigating carbohydrates bound to proteins. For example, deuterated disaccharides can be used as NMR probes to explore fucose-binding proteins.
DCL™ in Deuteration of Carbohydrates
Our DCL™ technology platform has developed new methods to synthesize important intermediates that play an important role in the preparation of deuterated drugs. In addition, new catalysts for inexpensive and easily scalable deuteration of various types of compounds and tools for in vitro drug sensitivity sensing have been developed. We can evaluate the degree of deuteration and selectivity achievable with various H/D substitution techniques, providing you with an adequate synthetic route.
Our custom solutions for deuterated carbohydrates, with high levels of regio- and stereoselectivity, a broad substrate range and mild reaction conditions, tolerate a wide range of functional groups and complex molecular structures, and are committed to providing customers with top-of-the-line service experience.
References
- Zhao G, et al. Excited-State Palladium-Catalyzed 1, 2-Spin-Center Shift Enables Selective C-2 Reduction, Deuteration, and Iodination of Carbohydrates. Journal of the American Chemical Society. 2021, 143(4): 1728-1734.
- Lowery R, et al. Deuterated carbohydrate probes as ‘label-free’substrates for probing nutrient uptake in mycobacteria by nuclear reaction analysis. Chemical Communications. 2015, 51(23): 4838-4841.
- Hellebrandt W, et al. Deuterated Disaccharides for the Investigation of Protein-Carbohydrate Interactions-Application of Bioaffinity-and STD-NMR. Journal of Carbohydrate Chemistry. 2000.
Customer Intellectual Property Protection
BOC Sciences has always regarded intellectual property as the most valuable asset of the company and its customers. We have signed non-disclosure agreements with customers and employees before the project starts, and provide synthetic route design and synthesis services in strict accordance with the terms of the non-disclosure agreement, striving to provide customers with target compounds in the shortest time possible.


 Fig.1 Schematic illustration for assessing 2H-carbohydrate uptake by M. smegmatis and ion beam analysis.[2]
Fig.1 Schematic illustration for assessing 2H-carbohydrate uptake by M. smegmatis and ion beam analysis.[2]

